|
5/20/2023 0 Comments Ion bonding force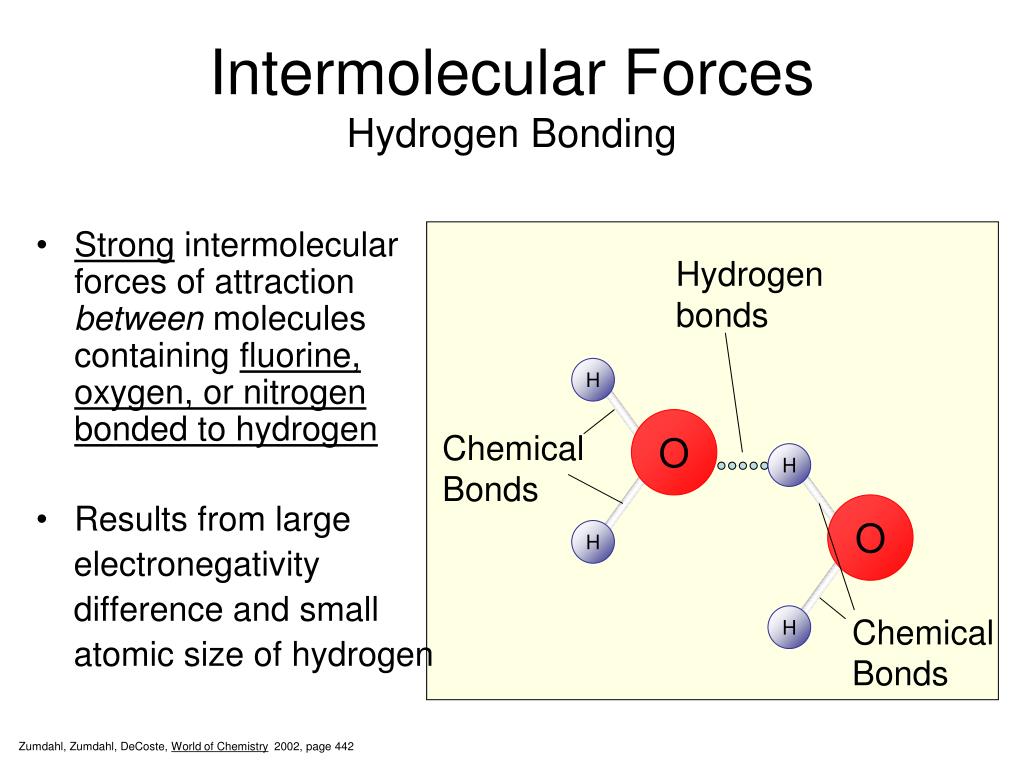 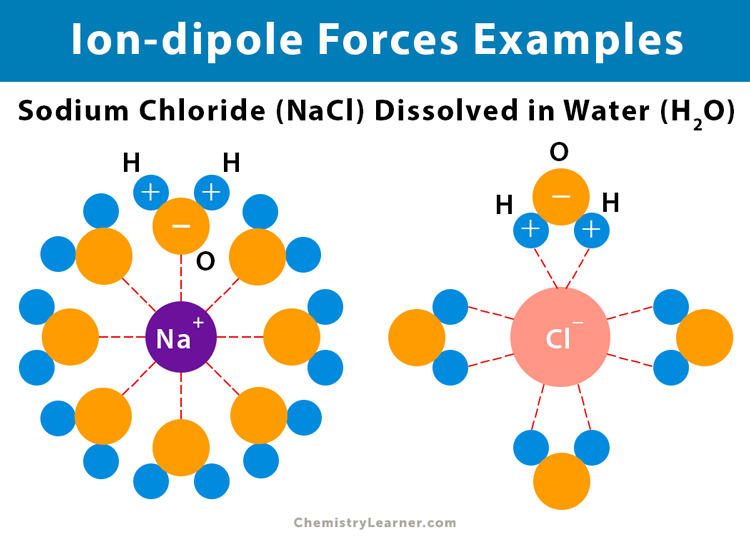
Similar to the covalent bond, the metallic bond involves electron sharing. In this molecule, hydrogen has a partial positive charge while fluorine has a partial negative charge. An example of a molecule with a polar covalent bond is hydrogen fluoride. When a polar covalent bond is formed, the result is an unequal sharing of electrons between atoms. A polar covalent bond exists when the electronegativity difference is somewhere in between, generally more towards the covalent side (small electronegativity difference). A large difference in the electronegativity of two atoms indicates a greater ionic character and is considered a purely ionic bond whereas a very small, negligible difference is considered a purely covalent bond. It is important to understand that all ionic compounds (compounds formed by ionic bonds) have some measure of electron sharing (covalent bonding) even though an ionic bond is not considered to be a type of covalent bond. This type of bond is common to organic compounds, where the atoms composing the compounds are non-metals.Ī polar covalent bond is bond that has a mix of ionic character and covalent character. Covalent bonds form between atoms that have similar electronegativity values. The atoms will share one of their outer electrons with the other, such that each individual atom effectively has a complete outer orbital. Consider two chlorine atoms each with 7 electrons in each respective outer shell. Generally, sharing electrons in such a fashion allows each atom involved to achieve a more stable electron configuration. The covalent bond is formed when adjacent atoms share valence electrons. Ionic bonds most frequently form between metals and non-metals. A difference of 1.7 in electronegativity values generally suggests that if a bond forms, it will be ionic. Ionic bonds form between atoms that vastly differ in their electronegativity values. 
The electrostatic or Coulombic attraction between oppositely charged ions is what is called the ionic bond. Meanwhile, Cl now has a net negative charge and is considered an anion. Due to the transfer of its electron, Na is considered a cation, with a net positive charge. As a result, the transfer of one electron from Na to Cl is favored because both atoms will achieve a more stable electron configuration (full outer orbital shells of eight electrons). Na has one valence electron (an electron in the outermost orbital shell that can take part in bonding) while Cl has seven valence electrons. Consider the example of sodium (Na) bonding with chlorine (Cl) to produce sodium chloride (NaCl), also known as table salt. The ionic bond is a result of an electron transfer from one atom to another. There is no transfer or sharing of electrons involved in a secondary bond. Secondary bonds are formed from the subtle attraction forces between positive and negative charges. Primary bonds are formed when the bonding process involves a transfer or sharing of electrons. Bonds can be separated into two types: primary bonds and secondary bonds. A bond may be defined as a force that holds groups of two or more atoms together, causing them to behave as a single unit. There are several types of bonds to consider when analyzing the chemical composition of a compound.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
